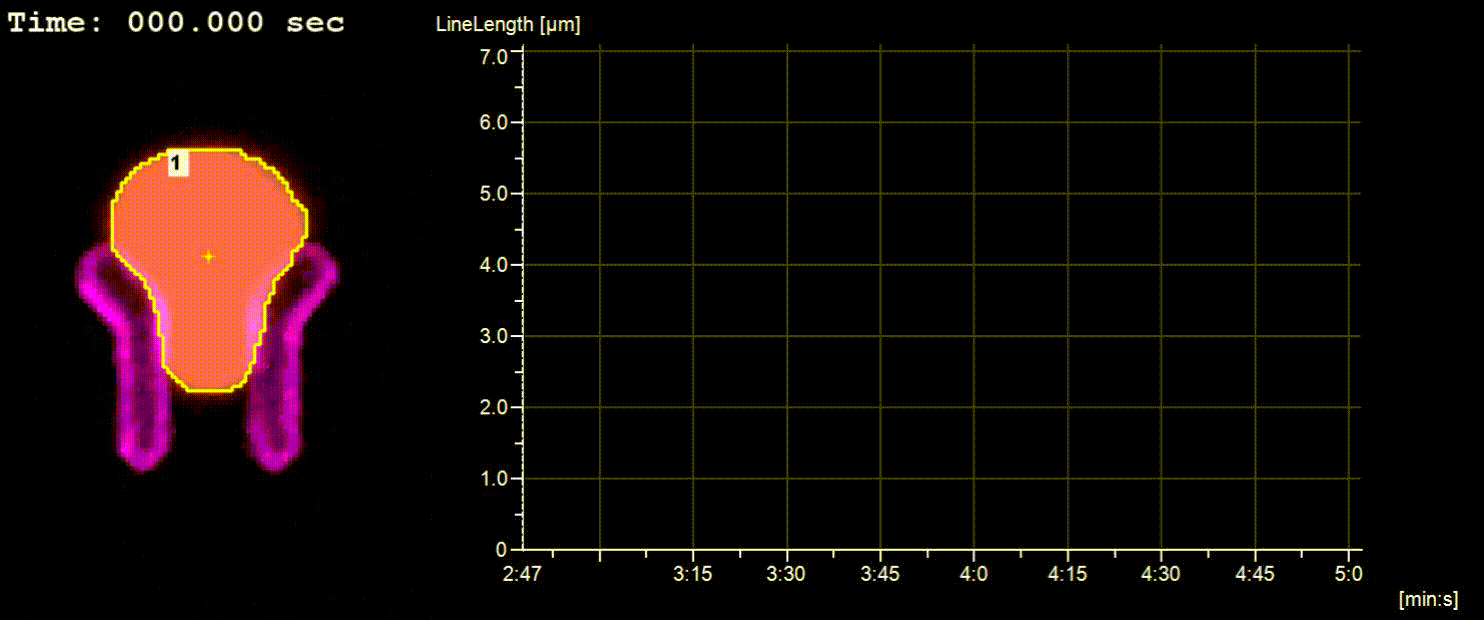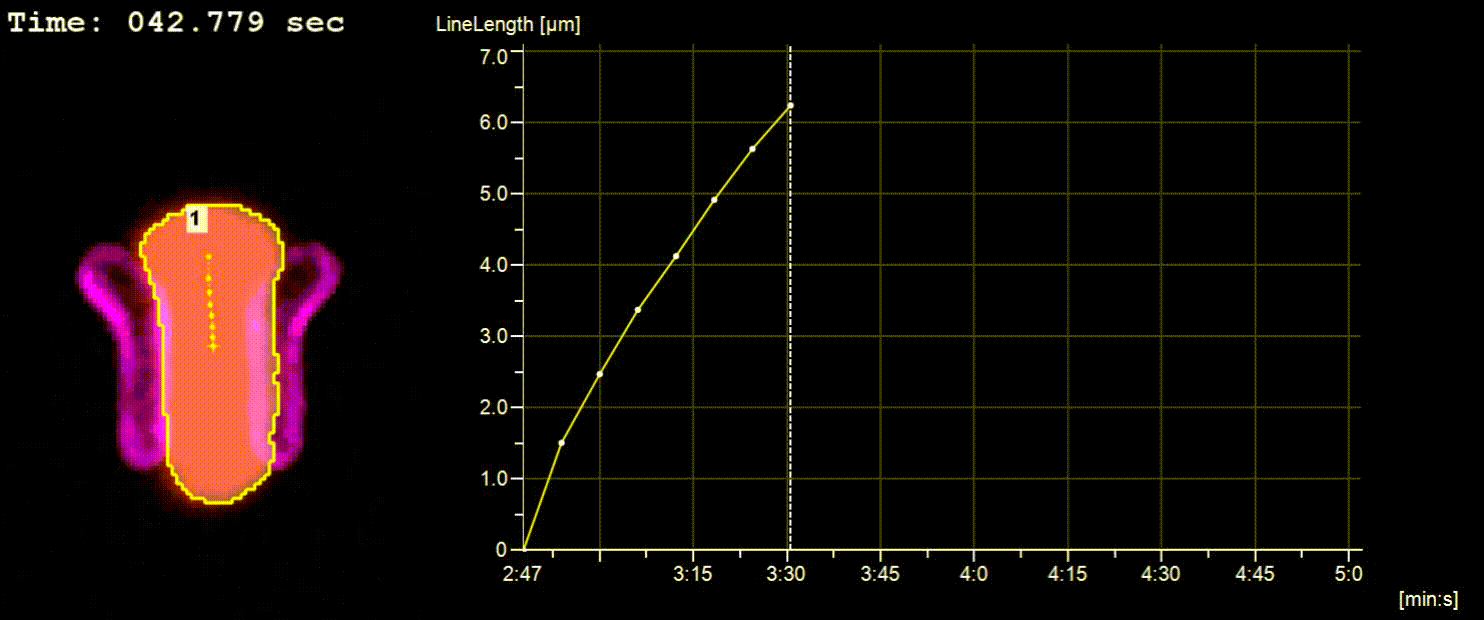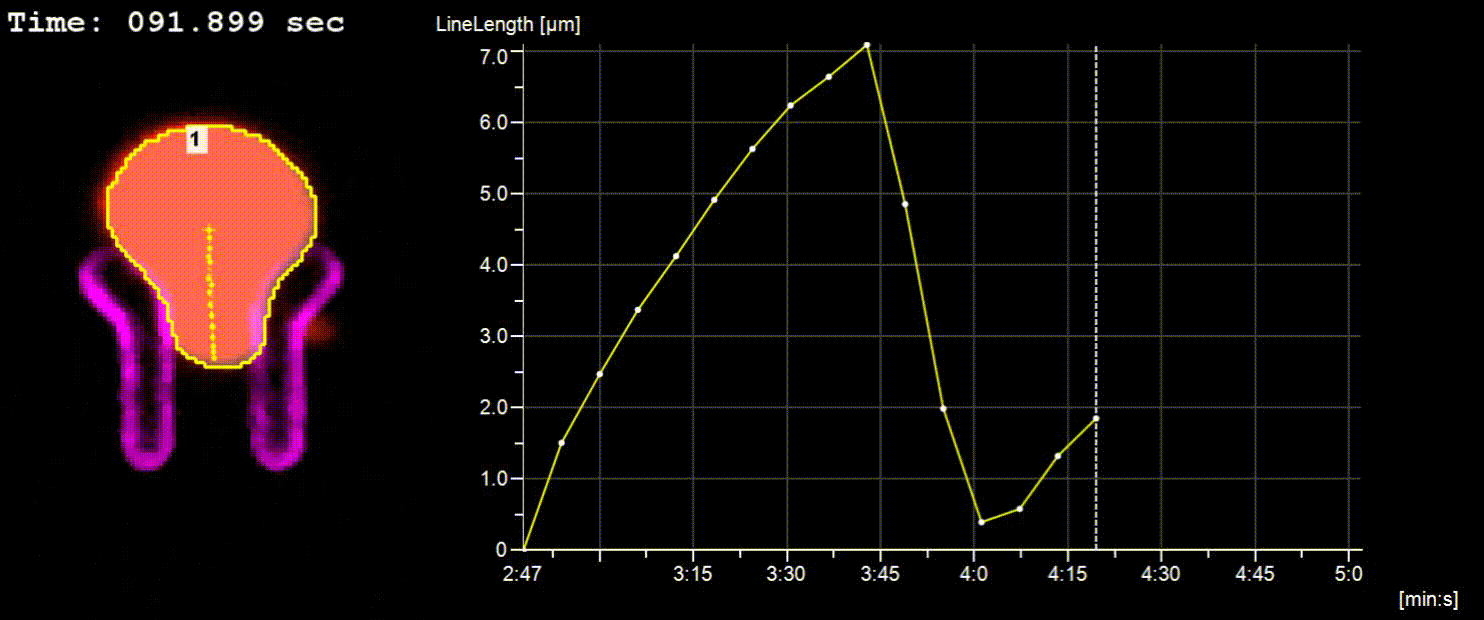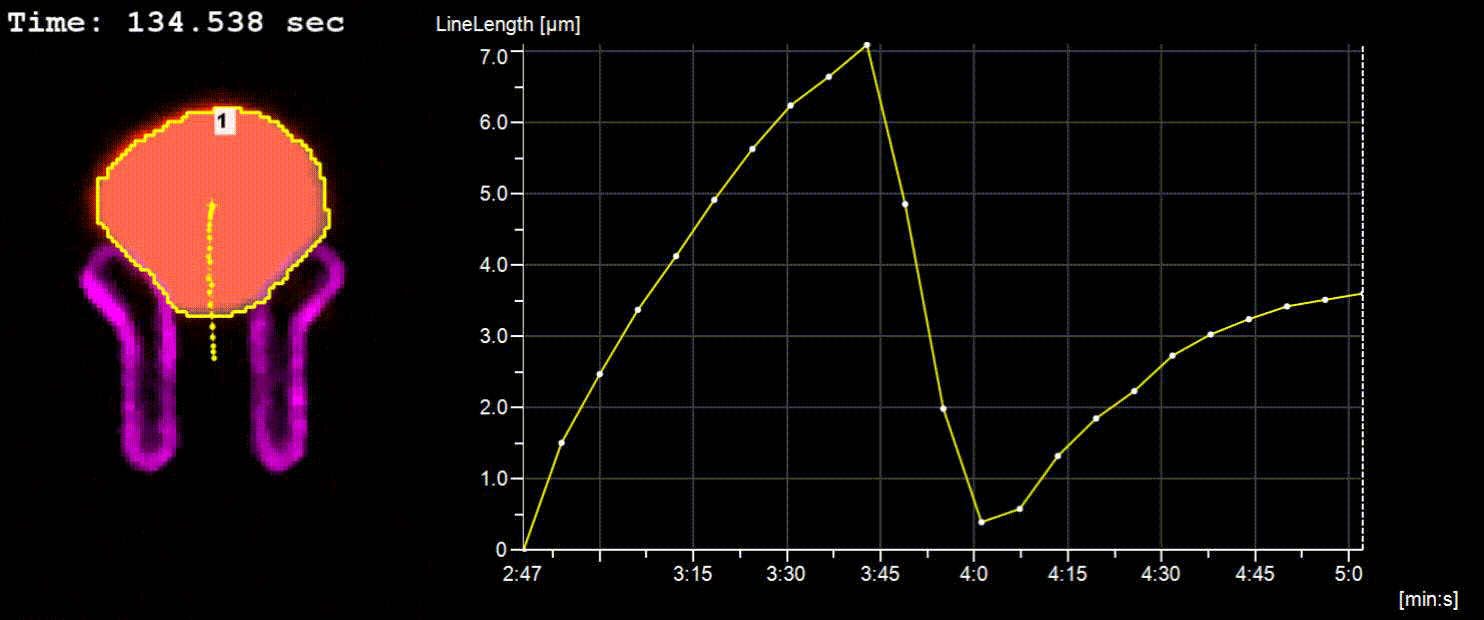
NucTrac Chip: Flow triggered nuclear rigidity measurements in a high density trap array
A flow triggered nuclear force measurement using a static nuclear trap array
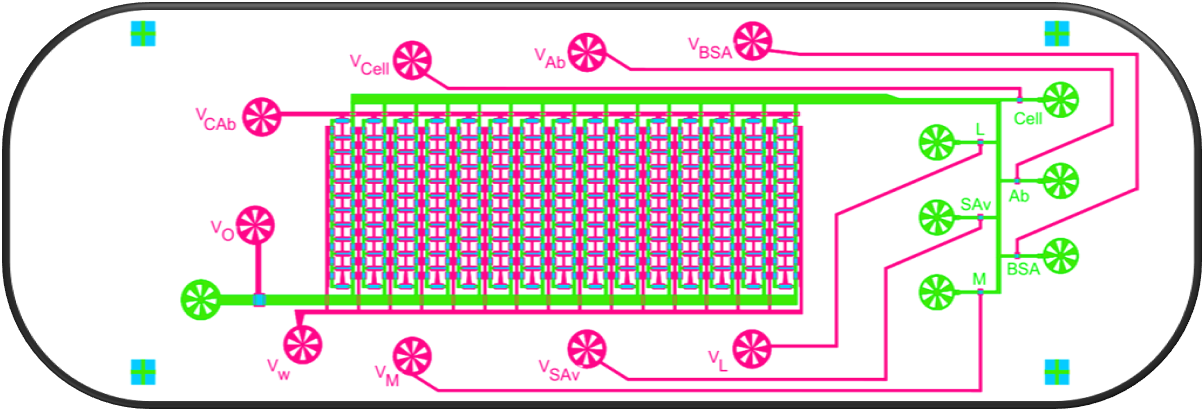
SiSec Chip: Microfluidic platform enables live-cell imaging of signaling and transcription combined with multiplexed secretion measurements in the same single cells

Innate immune cells, including macrophages and dendritic cells, protect the host from pathogenic assaults in part through secretion of a program of cytokines and chemokines (C/Cs). Cell-to-cell variability in C/C secretion appears to contribute to
the regulation of the immune response, but the sources of secretion variability are largely unknown.
To begin to track the biological sources that control secretion variability, we developed and validated a microfluidic device to integrate live-cell imaging of fluorescent reporter proteins with a single-cell assay of protein secretion. We used this device to image NF-κB RelA nuclear translocation dynamics and Tnf transcription dynamics in macrophages in response to stimulation with the bacterial component lipopolysaccharide (LPS), followed by quantification of secretion of TNF, CCL2, CCL3, and CCL5. We found that the timing of the initial peak of RelA signaling in part determined the relative level of TNF and CCL3 secretion, but not CCL2 and CCL5 secretion. Our results support evidence that differences in timing across cell processes partly account for cell-to-cell variability in downstream responses, but that other factors introduce variability at each biological step.
the regulation of the immune response, but the sources of secretion variability are largely unknown.
To begin to track the biological sources that control secretion variability, we developed and validated a microfluidic device to integrate live-cell imaging of fluorescent reporter proteins with a single-cell assay of protein secretion. We used this device to image NF-κB RelA nuclear translocation dynamics and Tnf transcription dynamics in macrophages in response to stimulation with the bacterial component lipopolysaccharide (LPS), followed by quantification of secretion of TNF, CCL2, CCL3, and CCL5. We found that the timing of the initial peak of RelA signaling in part determined the relative level of TNF and CCL3 secretion, but not CCL2 and CCL5 secretion. Our results support evidence that differences in timing across cell processes partly account for cell-to-cell variability in downstream responses, but that other factors introduce variability at each biological step.
Pop Slide Patterning : Fabricating glass slides with polymer microstructures
 Pop - slide patterning: Rapid fabrication of microstructured gaskets on glass
Pop - slide patterning: Rapid fabrication of microstructured gaskets on glass
We describe a rapid method of producing thin film microstructures on glass without using expensive equipment’s like UV lamp, plasma cleaner, ion etcher or a spin coater. Having designed to be exclusively simplistic, this low cost patterning method only requires a standard hotplate while allowing researchers from any domain to easily replicate features from an SU8 master mold within 45 minutes. The unique combination of heating temperatures along with a mixture of elastomer and releasing agent, allows us to easily produce microstructured gasket on glass. In addition, the thin film microstructures can be removed from the surface of glass and used as a membrane for mechanistic studies. Using this method we produced several microstructures such as micro-wells, micro-pillars, micro-gratings and microchannels with varying sizes (3 µm – 250 µm) and aspect ratios (0.066 – 3.8). Use of these thin film microstructures on glass makes it an impressive target for cell screening studies widely applicable in many research domains.
Passive Flow µFluidic Cell Docks
 Passive flow single cell traps
Passive flow single cell traps
Quantifying cell-to-cell variability in drug response dynamics is important when evaluating therapeutic efficacy. For example, optimizing latency reversing agents (LRAs) for use in a clinical ‘‘activate-and-kill’’ strategy to purge the latent HIV reservoir in patients requires minimizing heterogeneous viral activation dynamics. To evaluate how heterogeneity in latent HIV activation varies across a range of LRAs, we tracked drug-induced response dynamics in single cells via live-cell imaging using a latent HIVGFP reporter virus in a clonal Jurkat T cell line. To enable these studies in suspension cells, we designed a simple method to capture an array of single Jurkat T cells using a passive-flow microfluidic device. Our device, which does not require external pumps or tubing, can trap hundreds of cells within minutes witha high retention rate over 12 hours of imaging. Using this device, we quantified heterogeneity in viral activation stimulated by transcription factor (TF) activators and histone deacetylase (HDAC) inhibitors. Generally, TF activators resulted in both faster onset of viral activation and faster rates of production, while HDAC inhibitors resulted in more uniform onset times, but more heterogeneous rates of production. Finally, we demonstrated that while onset time of viral gene expression and rate of viral production together predict total HIV activation, rate and onset time were not correlated within the same individual cell, suggesting that these features are regulated independently. Overall, our results reveal drug-specific patterns of noisy HIV activation dynamics not previously identified in static single-cell assays, which may require consideration for the most effective activate-and-kill regime.
Jetting Microfluidics and DLD based cell sorting
 Concentration of cell encapsulated droplets using DLD
Concentration of cell encapsulated droplets using DLD
Activated proteases such as matrix metalloproteinases (MMPs) secreted from cancer cells can degrade the extracellular matrix (ECM) and contribute to tumour formation and metastasis. Measuring MMP activity in individual cancer cells can provide important insights on cancer cell heterogeneity and disease progression. Here, we present a microfluidic platform combining a droplet jetting generator and a deterministic lateral displacement (DLD) size-sorting channel that is capable of encapsulating individual cancer cells inside picoliter droplets effectively. Droplet jetting with cell-triggered Rayleigh-Plateau instability was employed which produced large droplets capable of cell encapsulation (diameter, 25 mm) and small empty droplets (diameter, 14 mm), which were then size-separated using a DLD size-sorting channel to enrich the single-cell encapsulated droplets (78%), regardless of the cell density of input sample solutions. The droplets containing encapsulated cancer cells were collected in an observation chamber to determine the kinetic profiles of MMP secretion and the inhibitory response in the presence of the drug doxycycline at the single-cell level to reveal their heterogeneous MMPs secretion activities
Pinch Flow Single Cell Kinase Signaling Assay
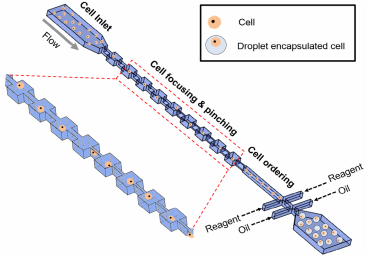 Single-cell kinase assay using droplet microfluidics
Single-cell kinase assay using droplet microfluidics
Droplet-based microfluidics has shown potential in high throughput single cell assays by encapsulating individual cells in water-in-oil emulsions. Ordering cells in a micro-channel is necessary to encapsulate individual cells into droplets further enhancing the assay efficiency. This is typically limited due to the difficulty of preparing high-density cell solutions and maintaining them without cell aggregation in long channels (>5 cm). In this study, we developed a short pinched flow channel (5 mm) to separate cell aggregates and to form a uniform cell distribution in a droplet-generating platform that encapsulated single cells with >55% encapsulation efficiency beating Poisson encapsulation statistics. Using this platform and commercially available Sox substrates (8-hydroxy-5-(N,N-dimethylsulfonamido)-2-methylquinoline), we have demonstrated a high throughput dynamic single cell signaling assay to measure the activity of receptor tyrosine kinases (RTKs) in lung cancer cells triggered by cell surface ligand binding. The phosphorylation of the substrates resulted in fluorescent emission, showing a sigmoidal increase over a 12 h period. The result exhibited a heterogeneous signaling rate in individual cells and showed various levels of drug resistance when treated with the tyrosine kinase inhibitor, gefitinib.
Droplet Microfluidics For High Throughput Single Cell Lysis
 Single cell lysis in Microdroplets
Single cell lysis in Microdroplets
One of the essential issues in cell biology is to analyze intracellular proteins that play an important role in signal transduction and processes. Standard assays for intracellular proteins are based on ensemble measurements which rule out the differences of intracellular protein expression in individual cells. Especially in cancer biology, only a few tumour cells turn into circulating tumour cells causing metastasis. In such cases, understanding the heterogeneity in cellular physiology would be necessary. To screen for intracellular protein expression among individual cells effectively, a reliable method to lyse individual cells in a high throughput manner is essential. Here, we demonstrate an integrated microflidic platform combining a cell focusing channel, reagent injection inlet and a droplet generator for high throughput single cell lysis. Individual cells are encapsulated into the droplets with chemical reagents which result in disrupting the cell membrane integrity, causing cell lysis. High throughput mammalian cell lysis was performed thus facilitating intracellular protein analysis on a micro device.
Microfluidic Bioluminescence based Receptor Quantification Assay
 Microfluidic Bioluminescence based Receptor Quantification Assay (MBRQA)
Microfluidic Bioluminescence based Receptor Quantification Assay (MBRQA)
The number of receptors expressed by cells plays an important role in controlling cell signaling events, thus determining its behaviour, state and fate. Current methods of quantifying receptors on cells are either laborious or do not maintain the cells in their native form. Here, a method integrating highly sensitive bioluminescence, high precision microfluidics and small footprint of lensfree optics is developed to quantify cell surface receptors. This method is safe to use, less laborious, and faster than the conventional radiolabelling and near field scanning methods. It is also more sensitive than fluorescence based assays and is ideal for high throughput screening. In quantifying β1 adrenergic receptors expressed on the surface of H9c2 cardiomyocytes, this method yields receptor numbers from 3.12 × 10^5 to 9.36 × 10^5 receptors/cell which are comparable with current methods. This can serve as a very good platform for rapid quantification of receptor numbers in ligand/drug binding and receptor characterization studies, which is an important part of pharmaceutical and biological research.
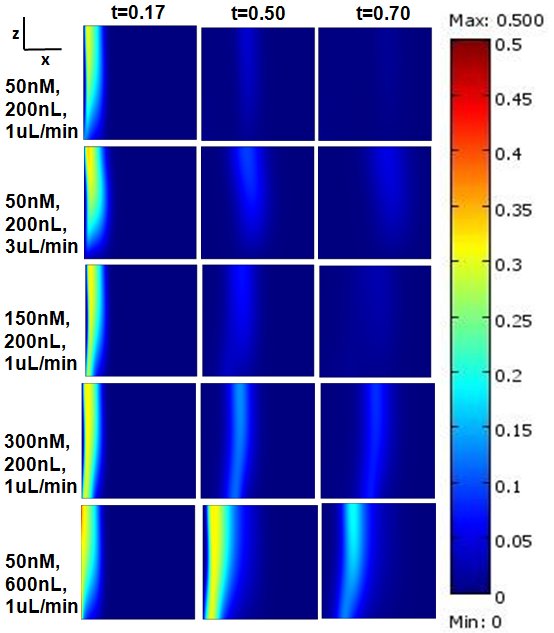
Microfluidic Displacement Assay
 Microfluidic Displacement Assay (MDA)
Microfluidic Displacement Assay (MDA)
The kinetic rate of dissociation of a ligand or drug binding to target cell surface receptors is an important parameter in biochemical research and drug design. Conventional methods of determining ligand dissociation rates suffer from serious limitations such as lack of in-vivo environment and mass transfer artifacts. We demonstrated a method to determine ligand dissociation rate from live cell surfaces using a microfluidic displacement technique, wherein a ligand is displaced from the surface receptors by an excess of a ligand analogue. A mathematical model describing transient unbinding and transport of the displaced ligand within the microchannel is presented here. This model allows simultaneous determination of the receptor internalization rate. We presented experimental data on the interaction between epidermal growth factor (EGF) and EGF receptors on the surface of A431 epidermoid carcinoma cells. Our experimental setup allowed us to evaluate the effects of incubation time, flow velocity and displacing ligand concentration on the measured rate constants. Additionally, we provide a graphical guide based on a mass transfer model to select the displacing ligand concentration that will prevent ligand rebinding.
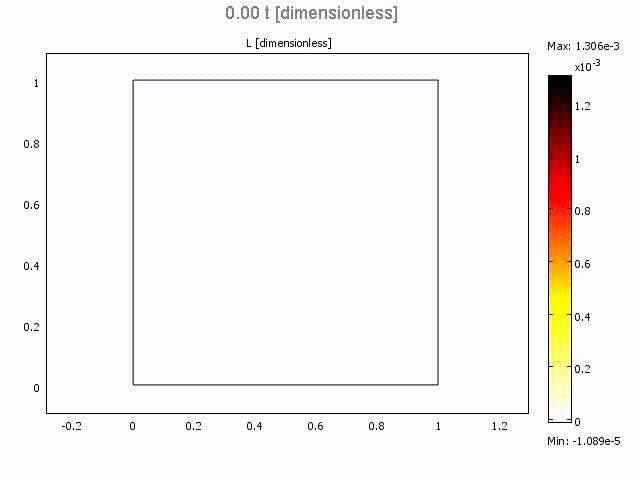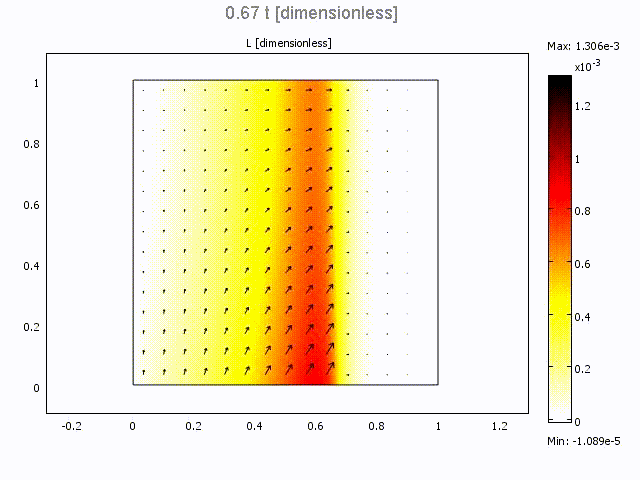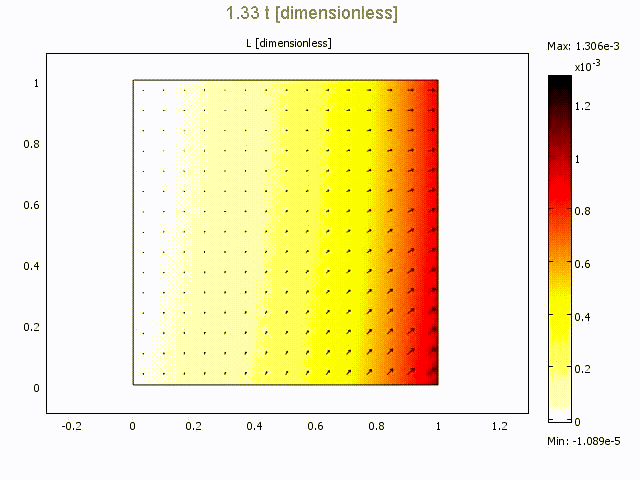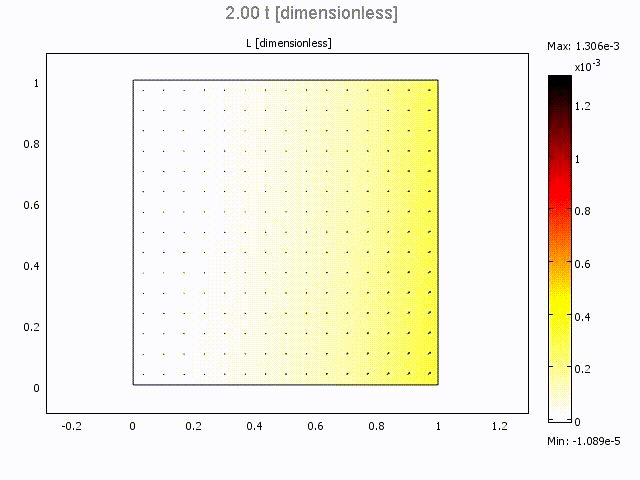
Microfluidic Bolus Induced Gradient Generator
 Microfluidic Bolus Induced Gradient Generator (BIGG)
Microfluidic Bolus Induced Gradient Generator (BIGG)
Major events in cell biology are initiated by the binding of ligands to cell surface receptors and/or their transport into cells. We present a study of a simple microchannel system that integrates a bolus generator and surface-adhered cell culture domains. Our system allows the delivery of small packets or boluses of biomolecules to a cell population. Owing to pressure driven microfluidic flow of the bolus, a gradient of cell surface bound ligands is established along the length of the microchannel. Experimental data for the epidermal growth factor (EGF) binding to its receptor on A431 cells are presented. We highlight the effect of changing Peclet number (or flowrate), bolus shape, bolus volume and ligand concentration on the gradient formed longitudinally in the microchannel. A mathematical model describing the transient convection, diffusion, dispersion and binding of ligands to cell surface receptors is developed. The model provides essential design guidelines for our system with good qualitative agreement with experimental data. The results suggest ways to modulate the amount of bound ligand and the gradient independently. This simple microsystem is suitable for generating longer range gradients involving larger cell populations as compared to existing microfluidic systems.